Last Updated on March 15, 2020
Cartilage is a group of strong and resilient tissues composed of chondrocytes surrounded by specialized mucopolysaccharide tissue.
It is found in many areas in the bodies including
- Joints
- Rib cage
- Ear
- Nose
- Bronchial tubes
- Intervertebral discs
It is much more elastic than bone but is stiffer and less flexible than muscle.
Types of Cartilage
Hyaline
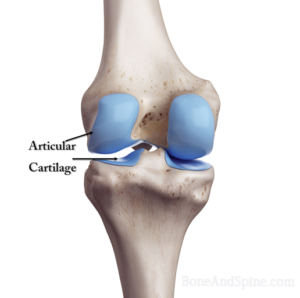
It is the type of cartilage we see in the joints and therefore called articular cartilage as it lines the joints. It is bluish-white and translucent due to very fine collagen fibers. Its distribution is most abundant and has a tendency to calcify after 40 years of age.
It is the most widespread type that is found in
- The articular surface of bones
- Forms the anterior portion of the ribs
- Support for the respiratory passages (ringlike structures of the trachea).
- Provides shape as in the nose
Hyaline cartilage is a flexible, elastic, bluish-white, and opalescent. Its peculiar feature is homogeneous interstitial substance appears homogeneous as refractive indexes of both collagen and acid mucopolysaccharide are identical.
Glycosaminoglycans, chiefly chondroitin sulfate, are contained within the interstitial substance and are responsible for the basophilic staining. matrix is primarily made of type II collagen and chondroitin sulphate.
Proteoglycans are responsible for the compressive stiffness of the tissue and its ability to withstand the load. The collagen provides tensile strength and resistance to shear.
Microscopically, there are chondrocytes in a group of two or more in a homogenous matrix. The cells are present in cavities called lacunae.
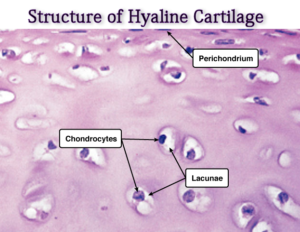
It has got no blood vessels except an occasional one passing through to other tissues. Its nourishment, depending upon the location could be from synovial fluid or perichondrium.
The perichondrium is the external covering of the articular cartilage. At the articulating surfaces, the synovial membrane is the outer covering. This contains blood vessels which provide nutrition.
Function
- Articular Cartilage ensures smoothness of joint movement and also acts as a shock absorber to some extent.
- It also stores synovial fluid like water in a sponge. on movement or weight-bearing, the synovial fluid is squeezed out, for lubrication.
- During the bone formation of the bones that undergo endochondral ossification, the framework is set by chondrocyte to form the cartilaginous structure which gets ossified.
Fibrocartilage
Fibrocartilage or fibrous cartilage differs from hyaline by the presence of thick, compact bundles of collagenous fibers within its interstitial substance. These bundles are arranged parallel to each other, separated by clefts in which encapsulated cells are squeezed.
Fibrocartilage appears to be a transitional tissue between hyaline cartilage and collagenous tissue and, as such, occurs in special places.
It can be called a specialized form of connective tissue in which the ground substance is cartilage. T
The cell types present are both fibrocytes and chondrocytes.
In contrast to the hyaline type, the matrix is largely acidophilic due to the presence of large numbers of collagen type I fibers. The ground substance between the fibers is basophilic and contains chondroblasts/cytes within lacunae.
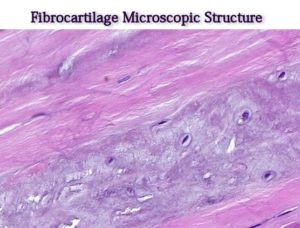
Fibrocartilage does not contain a perichondrium.
It is white and opaque due to the abundance of dense collagen fibers.
Wherever fibrous tissue is subjected to great pressure, it is replaced by fibrocartilage which is tough, strong and resilient.
Examples of fibrocartilage are
- Intervertebral disc
- Intra-articular discs
- Menisci
- Labrum [as in glenoid cavity and acetabulum]
- Lines certain bony grooves in which the tendons play.
Functions
- Absorb the shocks between the vertebrae in the spine.
- It provides sturdiness without impeding movement.
- It forms a firm joint between bones but still allows for a reasonable degree of movement.
- In articular cavities such as the ball-and-socket joints in the hip and shoulder regions, it deepens the sockets to make dislocation less possible.
Elastic Cartilage
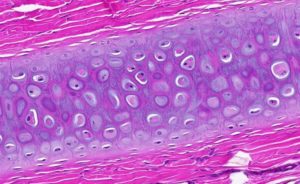
It is made up of numerous cells and a rich network of yellow elastic fibers pervading the matrix instead of collagen type 2. Therefore, it is more pliable. It does contain the perichondrium.
It is seen in
- External ear
- Auditory tube
- At the inlet of the larynx. It performs the following functions.
It maintains shape. In the ear, for example, it helps to maintain the shape and flexibility of the organ. It also strengthens and supports these structures.
Clinical Significance
Many diseases can involve cartilage. These are mainly
- Osteoarthritis
- Achondroplasia
- Costochondritis
- Spinal disc herniation
- Relapsing polychondritis – an autoimmune phenomenon
- Chondral tumors