Last Updated on March 14, 2020
Fracture healing in a broken bone is quite a complex process that begins by hematoma formation, a collection of inflammatory agents that render the bone-forming cells to form bone tissue to bridge the defect created by fracture.
In this article, we would discuss steps of fracture healing in a broken bone, the types of fracture healing and the factors that affect this healing process.
Relevant Anatomy and Physiology
From without inwards, the bone is composed of periosteum, cortex and medullary cavity.
The periosteum is a thick fibrous membrane covering the surface of the bone which consists of an outer fibrous layer, and an inner cellular layer. The cellular layer is osteogenic i.e. it can provide cells that cause new bone formation.
In fracture healing, the periosteum is the primary source of precursor cells that develop into chondroblasts ( cartilage cells) and osteoblasts (bone cells) during the repair.
The periosteum is tethered to the underlying bone by Sharpey’s fibers and is continuous with the capsule of the joint at the bone ends. The periosteum is rich in nerve fibers.
The cortex is made up of a compact bone which gives it the desired strength to withstand all possible mechanical stains.
Inside the cortical cylinder is the hollow called medullary cavity which is filled with red or yellow bone marrow.
When a bone breaks, it bleeds from its torn ends due to injury to its vessels which supply the blood.
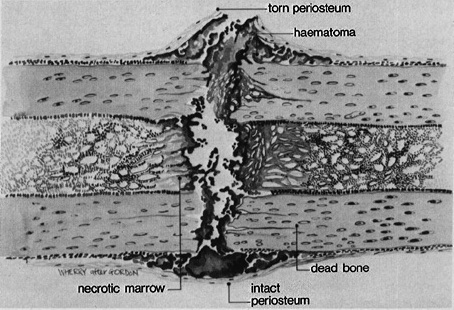
Injury to the vessels leads to the collection of the blood at the fracture site. The blood is collected at the fracture site. The collected blood is called fracture hematoma. [If a fracture is of open nature, then the blood comes out through the wound that communicates with fracture and hematoma is not formed. This is contemplated to be a major cause of poor bone fracture healing in open fractures.]
Inflammatory changes occur in the hematoma over the next few hours. The basic purpose of the inflammation is to contain the damage and facilitate healing and regeneration.
This inflammation brings in many cells that would help in the regeneration of the broken bone.
As time progresses, the fibroblasts ( cells that produce fibrous tissue) get interspersed with small vessels and form a loose mesh-like structure or granulation tissue uniting the broken ends of the bone.
Over the next few days, the cells of the periosteum replicate and develop into chondroblasts and osteoblasts and form woven bone that bridges the fracture. This bridge is visible on the x-ray at about three weeks and is called fracture callus.
The formation of callus is the first sign of union visible in x-ray and generally appears around 2-3 weeks after fracture. Eventually, the fracture gap is bridged by the cartilage and woven bone, restoring some of its original strength.

In the next section, we discuss this process of bone healing in detail.
Details of Fracture Healing and Different Steps Involved
This whole process of bone healing can be divided into three phases. These phases are
- Inflammatory phase
- Repair phase
- Remodeling phase
It must be understood that actual fracture healing is a continuous process and the events of different phases may overlap with respect to their occurrence.
That means events of the preceding phase may continue into the next phase and events of subsequent phases may begin in an earlier phase.
Inflammatory Phase
The bone breaks due to injury, along with its soft tissue envelope [periosteum and surrounding muscles].
The bleeding occurs due to the rupture of blood vessels crossing the fracture line. A hematoma is formed within the medullary canal, between the fracture ends, and beneath any elevated periosteum.
Due to loss of blood supply, immediate ends of fracture fragments undergo necrosis which leads to an immediate and intense acute inflammatory response.
The blood vessels dilate and there is an exudation of plasma to the injured site, further contributing to the swelling.
This brings acute inflammatory cells – macrophages, neutrophils, and platelets which release several factors such as
- Plasma-derived growth factor [PDGF]
- Tumor necrosis factor-alpha
- Absence [as in HIV infection] delays ossification
- Transforming growth factor-beta
- Interleukins L-1,6, 10,12
These factors are detected as early as 24 hours after injury.
Fibroblasts and mesenchymal cells migrate to the fracture site and granulation tissue forms around the fracture end followed by the proliferation of osteoblasts and fibroblasts.
Reparative Phase
The repair cells are of mesenchymal origin and are pluripotent cells [stem cells]. Mesenchyme is an embryonic connective tissue that is derived from the mesoderm. Pluripotent cells have the capacity to differentiate into bone, cartilage, and collagen.
The tissue formed eventually is determined by the microenvironment. High oxygen concentration and mechanical stability favor bone formation whereas low oxygen and instability lead to the formation of cartilage.
The repair cells produce the tissue known as callus, which is made up of fibrous tissue, cartilage, and young, immature bone. This quickly envelopes the bone ends and leads to a gradual increase in the stability of the fracture fragments.
Primary callus forms within two weeks. If the bone ends are not touching, then bridging soft callus forms.
Enchondral ossification converts soft callus to hard or bony callus, a type of woven bone. Medullary callus also supplements the bridging soft callus.
The amount of callus is dependent on the extent of immobilization.
As this phase of repair takes place, the bone ends gradually become enveloped in a fusiform mass of callus as noted in the picture above. Immobilization of the fragments occurs due to callus and is considered as one of the clinical signs of union.
Remodeling Phase
Remodeling about a fracture takes place for a prolonged period of time. In remodeling, osteoclasts resorb the woven bone trabeculae and new struts of bone are laid down that correspond to lines of force.
Remodeling is thought to be modulated by electrical signals. When a bone is subjected to stress, electro-positivity occurs on the convex surface and electro-negativity on the concave, a current produced by a piezoelectric effect. Regions of electropositivity are thought to be associated with osteoclastic activity and regions of electronegativity with osteoblastic activity.
The cellular module that controls remodeling is the resorption unit, consisting of osteoclasts, which first resorb bone, followed by osteoblasts, which lay down new Haversian systems.
Approximate Fracture Healing Time in Adults Taken by Different Bones
Depending on the fracture site, normal healing may take from 3-12 weeks.
- Phalanges fracture: 3 weeks
- Metacarpals fracture: 4-6 weeks
- Distal radius fracture: 4-6 weeks
- Distal humerus fracture: 8-10 weeks
- Humerus fracture: 6-8 weeks
- Femoral neck fracture: 12 weeks
- Femoral shaft fracture: 12 weeks
- Tibia fracture: 10 weeks
Types of Fracture Healing
Primary fracture healing and secondary fracture healing are two different types of healing in fractures depending on the rigidity of fixation of the fracture. The one described in the initial part of the article is a type of secondary fracture healing. But when the fragments are fixed rigidly by a plate or other gadget, the type of healing that occurs is primary.
Fracture stability dictates the type of healing that will occur.
Thus in surgically fixed fractures (fixed by plate and screws) where mechanical strain is minimal [less than 2%], primary fracture healing would occur.
If the strain is more [2%-10%], secondary bone healing will occur. In cases of the plaster cast or external fixator, secondary healing would follow.
Primary healing per se is rare and most of the fractures heal by secondary healing. This is so because management of most of the fractures allows some degree of movement.
Primary Bone Healing
In this kind of fracture, the healing callus is not formed at all and it occurs with rigid stabilization with or without compression of the bone ends. It is also called intramembranous healing and occurs via Haversian remodeling.
Rigid stabilization suppresses the formation of a callus in either cancellous or cortical bone.
Primary bone fracture healing can be divided into
- Gap healing
- Contact healing

Gap Healing
If internal fixation leaves a gap between fragments, the fracture heals by gap healing. Gap healing occurs in two stages.
Firstly, the width of the gap is filled by direct bone formation. An initial scaffold of woven bone is laid down, followed by the formation of lamellar bone as support. The orientation of the new bone formed in this first stage is transverse to that of the original lamellar bone orientation.
In the second stage, which happens after several weeks, longitudinal Haversian remodeling reconstructs the necrotic fracture ends and the newly formed bone to replace the woven bone with osteons of the original orientation. In the end, the normal bone structure results.
Contact Healing
Contact healing occurs where fragments are in direct apposition with [< 0.1 mm distance] and there is no interfragmentary strain so that osteons can grow across the fracture site, parallel to the long axis of the bone.
The process is initiated by osteoclasts forming cutting cones, that traverse the fracture line at 50-100 µm/day. This tunneling allows the penetration of capillaries and eventually the formation of new Haversian systems. These blood vessels are then accompanied by endothelial cells and osteoprogenitor cells for osteoblasts leading to the production of osteons across the fracture line eventually leading to regeneration of the normal bone architecture.
Secondary Fracture Healing
Secondary bone fracture healing occurs when there is no rigid fixation of the fractured bone ends, which leads to the development of a fracture callus. It includes an inflammatory phase, a reparative phase, and a remodeling phase as described above at the beginning of this article.
Secondary fracture healing occurs with non-rigid fixation, as fracture braces, external fixation, bridge plating, intramedullary nailing, etc.
Bone healing can occur as a combination of the above two processes depending on the stability throughout the construct.
Factors Affecting Fracture Healing
For bone healing to occur normally, the following requirements are to be met-
- The viability of fragments (i.e. intact blood supply)
- Mechanical rest – Immobilization by cast or fixation
- Absence of infection
Both local and systemic variables influence the rate and degree of fracture healing.
Age
Young patients heal rapidly and have a remarkable ability to remodel and correct angulation deformities. These abilities decrease once skeletal maturity is reached.
Nutrition
A substantial amount of energy is needed for fracture healing to occur. An adequate metabolic stage with sufficient carbohydrates and proteins is necessary.
It must be mentioned that while the lack of nutrition is detrimental to bone healing, there are no bone healing foods or food that could hasten the recovery of broken bones.
Systemic Diseases
Diseases like osteoporosis, diabetes, and those causing an immunocompromised state will likely delay healing. Illnesses like Marfan’s syndrome and Ehlers-Danlos syndrome cause abnormal musculoskeletal healing.
Calcium absorption is affected in gastric bypass patients leading to decreased Ca/Vit D levels, hyperparathyroidism (secondary) & increased calcium resorption from bone.
Diabetes mellitus affects the repair and remodeling of bone by decreasing the cellularity of the callus and delayed enchondral ossification.
HIV infection has a higher prevalence of fragility fractures with associated delayed healing.
Drugs
Bisphosphonates and systemic corticosteroids are associated with osteoporotic fractures on long-term use.
Studies have also implicated the use of NSAIDs affecting the healing adversely. Corticosteroids impede healing through many mechanisms.
Hormones
Thyroid hormone, growth hormone, calcitonin, and others play significant roles in bone healing.
Head Injury
It is a known observation that patients of head injury with associated fractures heal in shorter times because the head injury may increase osteogenic response.
Type of Bone
Cancellous or spongy bone fractures are usually more stable, involve greater surface areas, and have a better blood supply than do cortical (compact) bone fractures. Therefore, cancellous bone heals faster than cortical bone.
Degree of Trauma
The more extensive the injury to bone and surrounding soft tissue, the poorer the outcome. Open fractures let the hematoma escape into the soft tissues and may be associated with bone healing problems.
Blood Supply
Inadequate blood supply to the injured bony regions impairs healing. Some vulnerable areas are the femoral head, talus, and scaphoid bones.
Degree of Immobilization
The fracture site must be immobilized for blood vessel ingrowth and bone healing process. If there are repeated disruptions of repair tissue, the bone healing may be affected. Normal apposition of fracture fragments is needed for the union to occur. Inadequate reduction, excessive traction, or interposition of soft tissue will prevent healing.
Intraarticular Fractures
Synovial fluid contains collagenases that retard bone healing. The joint movement may cause the fracture fragments to move and thus affect union.
Infection
Infections cause necrosis and edema and take energy away from the healing process and may increase mobility of fracture site.
Local Pathologies
Local lesions of the bone may cause poor fracture healing. Some of the common reasons to cause poor healing are
- Primary and secondary malignancy
- Paget disease
- Fibrous dysplasia
- Irradiation
- Bone necrosis
Type of Fracture
The morphology of the fracture will suggest the type of injury that caused it, whether transverse, short oblique, or spiral, displaced or undisplaced, comminuted or not.
Read more about different types of fractures
Transverse and short oblique fractures are indicative of a greater force than are spiral fractures. Marked comminution and gross displacement, suggesting complete disruption of the soft tissue, emphasize the violent nature of the injury.
Impaired Fracture Healing
Normal fracture healing can be disrupted in numerous ways:
Delayed Union
Fracture healing takes about twice as long as expected for a specific location.
Non-union
Fracture healing does not occur within 6-9 months. Sites with known predilection are scaphoid bone, femoral neck, tibial shaft.
Malunion
It means healing of the fractures in the wrong position. It may or may not be compensated to a certain degree by remodeling of the bone.
Read More| Use of bone stimulator for fracture healing
References
- Balga R, Wetterwald A, Portenier J, et al. Tumor necrosis factor-alpha: alternative role as an inhibitor of osteoclast formation in vitro. Bone. 2006;39(2):325–35.
- Dimitriou R, Tsiridis E, Giannoudis PV. Current concepts of molecular aspects of bone healing. Injury. 2005;36(12):1392–404.
- Tsiridis E, Upadhyay N, Giannoudis P. Molecular aspects of fracture healing: which are the important molecules? Injury. 2007;38(Suppl 1): S11–25